|
By comparison, air molecules at room temperature, with an average speed of 2,000 mph, can have speeds anywhere between 0 and 4,000 mph. Remarkably these “supersonic beams” are nearly monoenergetic, meaning that the speeds of molecules will all be very close to the average: for example, if a beam comes out at 2,000 miles per hour, molecules in it will deviate from that speed by at most 20 mph. 
More than 40 years ago chemists found out that at a pressure of several atmospheres, gas escaping through a small hole into a vacuum undergoes significant cooling as it expands. 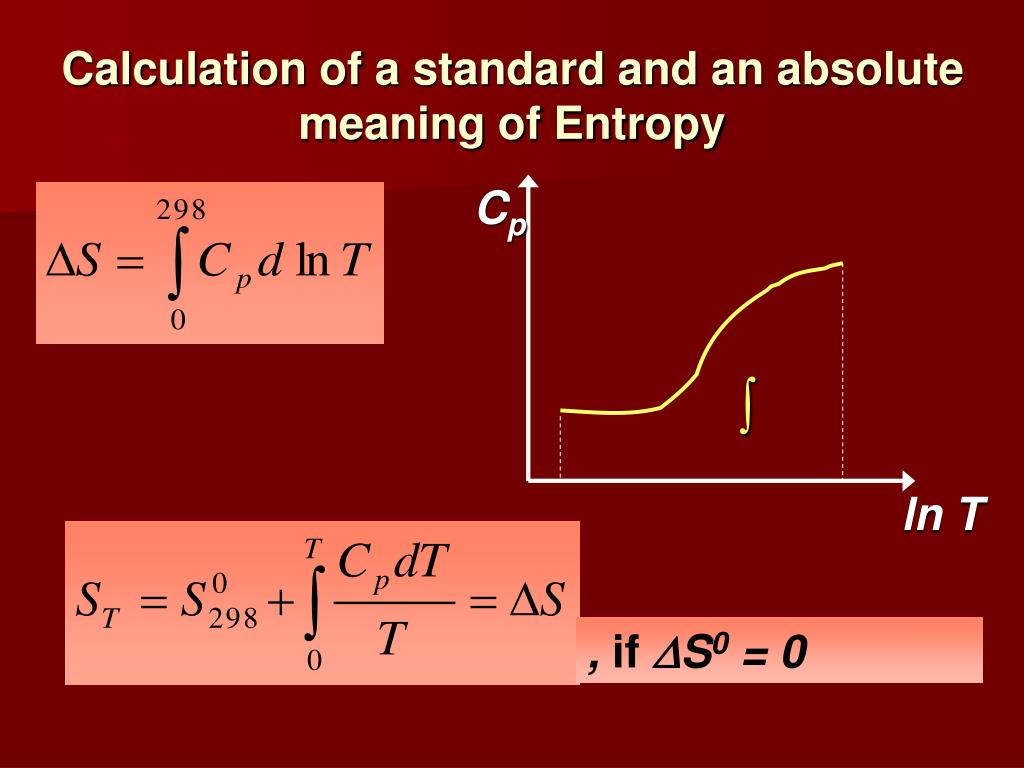
The gas must then be slowed, confined in a vacuum chamber and kept away from its walls. In a typical experiment, researchers begin by producing a rarefied gas of a certain chemical element by heating up a solid or vaporizing one with a laser. Stopping and manipulating atoms and molecules is no easy feat. And supercooling hydrogen and its isotopes could help small laboratories to answer questions in fundamental physics of the type that have traditionally required huge experiments such as those at particle accelerators. On the science side, cooling atoms and molecules may enable researchers to explore the no-man’s-zone between quantum physics and ordinary chemistry or to uncover possible differences in behavior between matter and antimatter. Another spin-off might be an increase in the precision of nanoscale fabrication methods that are used to make computer chips. For example, variants on the technique may lead to processes for purifying rare isotopes that have important uses in medicine and in basic research. The newfound capability will open directions in basic research and lead to a wide range of practical uses. This great Scottish physicist theorized the possibility of a “demon” that seemed able to violate the rules of thermodynamics. My inspiration: James Clerk Maxwell’s Victorian-era thought experiment. Now, however, my research group has demonstrated a new cooling method that works on most elements and on many types of molecules as well. For example, hydrogen, the simplest of all atoms, was for a long time extremely challenging to cool. The drawback of these atom-cooling techniques is that they are applicable to only a few of the elements in the periodic table, limiting their usefulness. In particular, cooling gaseous clouds of atoms-as opposed to matter in the liquid or solid state-to a small fraction of a degree above absolute zero has enabled researchers to observe matter particles behaving as waves, to create the most precise measuring instruments in history, and to build the most accurate atomic clocks. In such extreme realms, weird quantum effects begin to manifest themselves and to produce new and unusual states of matter. Nothing in nature is ever perfectly still, and the faster things go, the more energy they carry the collective energy of atoms and molecules is what we call, and feel as, heat.Įven though total stillness, corresponding to the temperature of absolute zero, is physically impossible, scientists have edged ever closer to that ultimate limit. Meanwhile the atoms and molecules that make up your body incessantly tumble, vibrate or collide with one another. 
As you read these words, the air’s molecules are zipping around you at 2,000 miles per hour, faster than a speeding bullet, and bombarding you from all sides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
